|
2/10/2024 0 Comments Elemental oxygen chargeSince it has the prefix per- and the suffix -ate, it must have one O atom more than chlorate, meaning the formula is ClO 4 –. Let’s look at the chart above and see if we can figure out what perchlor ate would be. Once you know the polyatomic ions that end in the suffix -ate there are only a few more patterns to know and you’ll be on your way to naming dozens and dozens of polyatomic ions! Increasingįor example, let’s use chlorate as an example. In group 15/VA, P and As have the same pattern, but notice N is different, combining with only three oxygens and having only a 1- charge. In group 16/VIA, both S and Se have the same pattern. Cl combines with three oxygens and has a 1- charge Br combines with three oxygens and has a 1- charge I combines with three oxygens and has a 1- charge. For example, if you know that chlorate is ClO 3 –, then you can predict that all of the other oxyanions made from nonmetals in group 17/VIIA follow the same pattern. Within a column, the pattern also repeats.On the top row, the farthest right polyatomic -ate ion is nitrate which is 1-, followed by carbonate which is 2-, followed by borate which is 3. The second column (sulfate, selenate) all have a 2- charge, while the third column (phosphate, arsenate) all have a 3- charge. As you start from right-hand side, the first column of polyatomic -ate ions (chlorate, bromate, iodate) all have a 1- charge.Notice how the four polyatomic -ate ions in the center square (phosphate, arsenate, sulfate, and selenate) all have four oxygen atoms, while the polyatomic -ate ions on the outside all have three oxygen atoms.However, the number of oxygens and the charge on the ions varies as we can see in the following table. These nonmetals for polyatomic ions that end in the suffix -ate. If you refer to the periodic table below, you will that a several nonmetals have been highlighted that commonly form such oxyanions. Many of the nonmetals combine with oxygen to make anions (negatively charged ions) that we are sometimes referred to as oxyanions. The good news, is there are dozens of other polyatomic ions that are relatively easy to figure just by knowing or observing a few patterns. When it comes to memorizing the, flashcards are your friends! Cation Depending on your course or instructor, you may need to memorize some of the following. There are some rules and patterns which can help us to deduce the structures of these ions, but before we can learn those, there are a handful that don’t follow the patterns that you just need to memorize. (The prefix poly- means “many,” as in polygon or polytheism.) 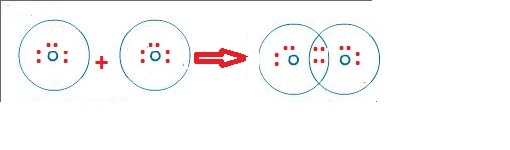 
Unlike monoatomic ions, which are composed of a single atom, polyatomic ions are composed of many atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
